Defective Products and Physiomesh
More than 2,000 people have court actions pending against Ethicon because of complications related to their Physiomesh hernia repair patches. Ethicon is a medical device company owned by Johnson & Johnson. The courts consolidated the cases into multidistrict litigation, where the case is ongoing as of July 2019.
If you had complications or required revision surgery following a hernia repair procedure utilizing Physiomesh, Berger and Green want to review your case. You may be eligible to pursue compensation for your medical care, lost wages, pain and suffering, and more. Call 412-661-1400 for your free case review.
40+ years of experience from strong, knowledgeable, compassionate attorneys.
Start A Free EvaluationPatients Who Underwent Physiomesh Hernia Repair Allege the Mesh Is Defective
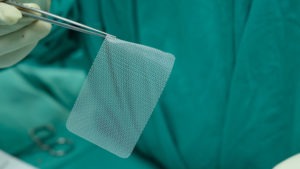
Ethicon Physiomesh is a patch often used to repair abdominal hernias. In theory, the product provides a stronger closure of the hernia than a traditional closure created by stitching the tissue closed. However, thousands of people whose surgeons used Physiomesh during their abdominal hernia repairs allege that the patches cause other serious complications and side effects. This includes:
- Causing chronic pain in the area
- Shrinking, migrating, and otherwise failing
- An increased risk of infection
- Difficult or impossible removal
When a Physiomesh patch fails or becomes infected, it often requires revision surgery. This is usually a more invasive, riskier procedure than the initial hernia repair.
A 2016 study team stopped their comparative study pitting Physiomesh against competitor Ventralight mesh after the researchers found that a full 20 percent of those who underwent a Physiomesh repair required additional treatment within six months. Surgical Endoscopy published the study.
Still, little has been done to hold Ethicon responsible. In 2015, Ethicon announced that it was withdrawing some Physiomesh products from the marketplace. However, the U.S. Food and Drug Administration has never recalled Physiomesh for any reason related to recurrence and reoperation rates.
We know you’re hurting. We can help. Free case evaluations, home and hospital visits.
Contact Us Now For HelpBerger and Green Can Explain Your Rights and Review Your Eligibility
At Berger and Green, our dangerous and defective device attorneys understand how frustrating and stressful it can be to undergo a procedure and end up in more pain than you were before the surgery. If your surgeon used Ethicon Physiomesh during your procedure, and you later suffered complications or require revision surgery, you may be able to pursue compensation.
The Berger and Green team is currently reviewing Physiomesh lawsuit cases nationwide. We can review your case at no cost to you, and you owe us nothing unless we recover a payout on your behalf. Call or text our attorneys today at 412-661-1400 to learn more.
We can build a strong case to prove your chronic pain and other complications cause you significant pain and suffering, as well as other losses. Let us go after the damages you deserve.
You need an attorney with the experience and dedication to give your case the care it deserves.
Start A Free EvaluationDamages Available in a Dangerous Medical Device Lawsuit
When we take action on your behalf in a suit or claim against Ethicon, the goal is to hold the medical device manufacturer accountable for making and marketing Physiomesh. Medical device companies have a responsibility to the general public to only market safe devices and to warn of all potential dangers. We allege that Ethicon failed to do so, leaving you in pain and requiring additional surgery.
To this end, the types of damages we may be able to recover from our lawsuit or claim against Ethicon in a Physiomesh case include:
- Medical care costs related to your complications
- Ongoing care costs, including pain management if necessary
- Lost wages and benefits
- Out-of-pocket costs
- Pain and suffering
- Punitive damages
We will help you collect proof of your related expenses, damages, and losses, and fight for a payout that covers all your economic and noneconomic damages. We can also pursue punitive damages in some cases.
We can address all the legal hurdles that may be keeping you from getting a fair settlement.
Speak To An Attorney TodayCurrent Status of the Ethicon Physiomesh Lawsuits
As of July 2019, the more than 2,000 lawsuits filed against Ethicon and their parent company Johnson & Johnson are consolidated into MDL 2782. In June of 2017, the Judicial Panel on Multidistrict Litigation formed all federal lawsuits related to Physiomesh into a single multidistrict litigation (MDL). MDL 2782 is still underway in the Northern District of Georgia.
Multidistrict litigation allows the plaintiffs to manage the discovery process and navigate all pretrial motions together, ensuring each plaintiff does not have to try to pay for their own investigation against a major international corporation. The court will also hear several bellwether cases to determine how a jury will react to the evidence each side presents.
Following the bellwether trials, all remaining cases will return to their original jurisdictions for settlement or trial.
Let Berger and Green review your case today. We may be able to take legal action on your behalf. Call or text us at 412-661-1400 to get started.
Talk to a Physiomesh Lawsuit Lawyer
If you underwent hernia repair surgery with a hernia patch and later required revision surgery — or if you suffer chronic pain or have other significant complications — you may be eligible to pursue a case against the manufacturer of that mesh patch. Ethicon Physiomesh is just one type of mesh hernia patch that patients link to serious complications and recurrence.
The dangerous medical device attorneys from Berger and Green can help you learn the type of patch used in your hernia repair and pursue compensation from Ethicon or another manufacturer on your behalf. We can document your related losses and expenses, and collect evidence to prove how much you suffered because of this hernia repair patch failure.
To get started today or to learn more about your rights after a Physiomesh hernia repair failure, call or text us today at 412-661-1400.






